What’s inside:
Patient safety is at the heart of every healthcare provider’s mission, and one of the highest areas of risk is in operating room (OR) and procedural rooms.
The point of care is fast-paced and unpredictable, so having robust procedures in place that lower risk is vital.
Despite this, healthcare providers regularly struggle with one specific task that can have a big impact on patient safety – the recording of surgical supply items used in surgery.
This blog looks at how new technology can boost patient safety at the point of use. It covers:
- The surgical documentation burden placed on nurses
- Pre consumption item validation
- The documentation of consumed items
- Recall management
- Using image-recognition and AI technology to improve surgical supply utilization
Unburdening nurses: Focusing on patients, not paperwork
Recording usage is a key task for the circulating nurse as it has operational, clinical and regulatory benefits – all of which underpin healthcare performance.
Point of use data capture technology in the surgical setting needs to serve three purposes:
- To validate the safety of every item prior to consumption
- To document every product used in surgery on the patient file
- To ensure digital documentation of all implants in case of a future recall
We will look at each of these in turn:
Item validation prior to consumption
Validation of items before consumption is crucial for patient safety as it helps prevent the use of counterfeit, expired, or recalled medical devices. Failing to validate items can lead to the following risks:
- Ineffective Treatment: “The use of unvalidated or faulty devices in surgery can compromise patient outcomes and the effectiveness of the treatment.” (Study: Journal of Surgical Research)
- Surgical Complications: “Unvalidated items can increase the risk of surgical complications, including infections, device failures, and adverse reactions.” (Study: Journal of Patient Safety)
- Adverse Events: “Not validating items prior to usage can contribute to adverse events, patient harm, and even fatalities.” (Study: Annals of Surgery)
The point of care is the last line of defense for identifying unsafe items prior to consumption. Technology can play a crucial role in helping nurses to notice items that should not be used in procedures.
NOTE! Having an efficient item validation process prior to consumption protects patients.
Full, accurate documentation of items used in surgery
Hospitals and surgery centers tend to use barcode scanning or manual inputting to record the items used in surgery.
System inefficiency: Some items simply won’t scan into the system. It could be that these items have changed coding that doesn’t match the entry on the Item Master. It may be that the item is not a stock item and therefore there is no match on the system. The reliance of barcode scanning and manual data entry on the Item Master means that the item identification process cannot be completed unless the updated details are corrected or the new item is enrolled – all of which cannot take place during surgery.
Time Constraints and Workload: The difficulty that nurses face recording usage means that they are focusing on product packets and data capture tools rather than patients.
According to a study by The Journal of PeriAnesthesia Nursing,
“Nurses often face time constraints and heavy workloads, making it challenging to thoroughly validate each item before usage.”
These manpower and technological limitations put patients at risk:
- Good practice is for every item to be documented during the procedure.
- Evidence suggests that delaying the documentation of products used in procedures runs the real risk of them never making it on to the system at all.
- Because documentation is time consuming when using inefficient data-capture tools, nurses are taken away from direct patient care to undertake supply chain admin – at the expense of focusing on patients.
NOTE! Nurses need a simple way to record every product used in surgery and can cope with a variety of items – even substitute items, trunk stock, or products that have an out-of-date listing on the Item Master. In addition, image recognition sensors can digitalize hand-written implant sheets, ensuring that every reportable item ends up on the patient file.
Recall Management – the need for digital data
Regulatory bodies, such as the Food and Drug Administration (FDA), have stringent requirements for tracking and documenting implants at the point of use. Regulations such as Unique Device Identifier (UDI) aim to ensure patient safety, traceability, and accountability throughout the implant’s lifecycle. Accurate digital documentation is necessary to comply with these regulations and maintain a high standard of care.
Inaccurate records can significantly hinder recall management in healthcare. When a product recall occurs, it is crucial to identify the affected patients promptly and take appropriate actions, such as notifying them, scheduling follow-up examinations, or arranging for replacement surgeries.
Accurate surgical documentation enables healthcare providers to efficiently track and locate patients who may have received recalled implants – this mitigates potential risks and ensures timely interventions.
It’s clear that traditional methods of documenting usage data in procedures are often not up to the task and threaten patient safety.
New technology, however, is now available that ensures quick and accurate usage documentation – and this is proving to be a gamechanger.
NOTE! Achieving full, accurate and digital surgical records on the EMR protects patients in the event of a future product recall.
Snap & Go – image recognition technology at the point of care
Snap & Go uses computer vision, AI and machine learning technologies to revolutionize medical device tracking – significantly improving patient safety while benefiting nurses and healthcare facilities.
Snap & Go is very simple to use, in fact it’s just like scanning an item on a self-check-out at the supermarket.
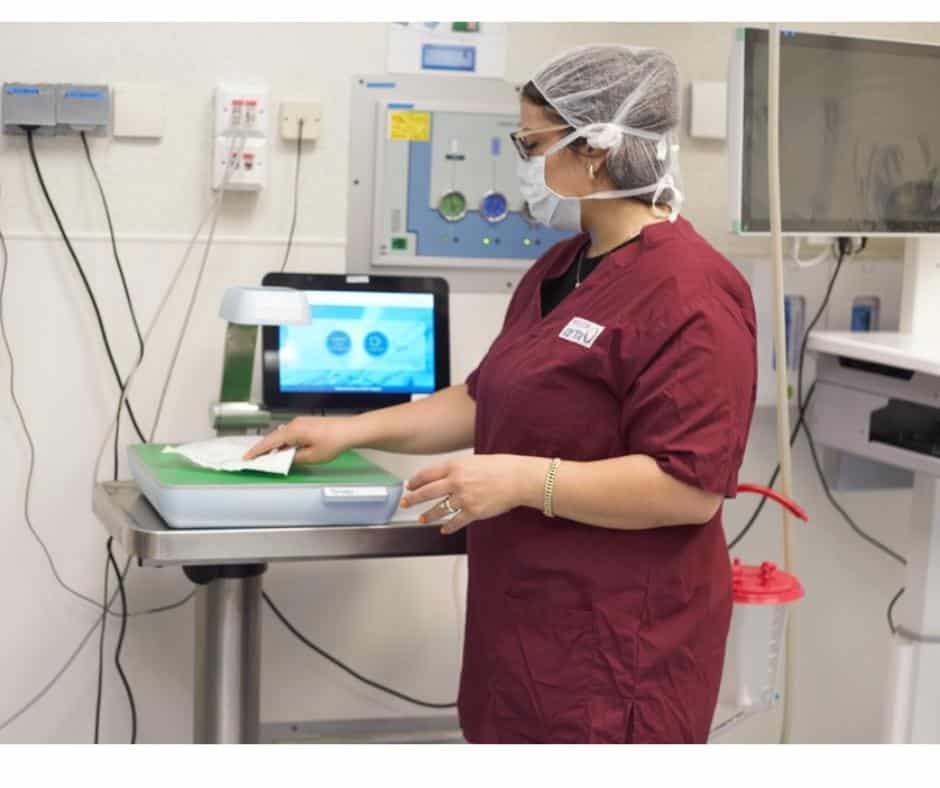
Nurses just need to take a quick ‘snap’ of the product packaging and the powerful system will then automatically identify the item, check for any product safety issues and then document it on the patient file in the EHR.
The nurse’s role in supply chain admin at the point of care is reduced to just 3 seconds per item! Hear what nurses have to say about Snap & Go.
Let’s look at how it tackles the common surgical documentation challenges that healthcare providers face.
Pre-consumption alerts: The last line of defense
Snap & Go’s has built-in safety features that proactively check every item selected for surgery.
The system quickly captures and identifies each item and flags up a pre-consumption recall and expiry alerts on the screen to prevent the nurse from handing the product over to the surgical team.
Product invalidity alerts act as a crucial safety net, preventing the accidental consumption of unsafe products.
These alerts serve as the last line of defense, ensuring that only validated and safe medical devices are used on patients.

Rapid recall management and reduced litigation
When digital records are complete and correct recall management just requires a simple system report.
By ensuring accurate and efficient tracking of medical devices, Snap & Go enables healthcare providers to swiftly respond to product recalls.
Recall management becomes a proactive task rather than a long, panicked reactive exercise.
Getting recall management right protects the healthcare organization against potential legal liabilities.
According to a report published by the American Society of Healthcare Risk Management,
“The implementation of comprehensive tracking systems can significantly reduce litigation costs by up to 50%.”
Snap & Go offers the simplest and most efficient item tracking capability on the market through its use of computer vision adn AI technology.
Empowering nurses – protecting patients
Snap & Go empowers nurses by simplifying their workflows and enabling them to focus on patient care rather than administrative tasks. As one nurse shared, “Snap & Go has transformed the way we work. It gives us more time to connect with patients and provide the quality care they deserve.”
Snap & Go protects patients, supports nurses and mitigates organizational risk.
Check out our 30 second video summary of how Snap & Go supports enhanced patient safety at the point of care.
If your healthcare provider is looking to boost patient safety at the point of care, contact us for a demo or to find out more.








